
A Pilot Study to Evaluate a Therapeutic Shoeing System for Laminitis Using a Steward Clog and Hoof Cast
Hircock1, FdSc AWCF; J. Mather1, BSc (Hons) AWCF; M. Caldwell2*, PhD FWCF
1The School of Veterinary Nursing and Farriery, Myerscough College, Billsborrow, Preston, PR3 0RY, UK
2Scientific Horseshoeing, 116, Newcastle Road, Talke, Staffordshire. ST7 1SA
*Corresponding author: info@hoofflix.com
Keywords: equine, laminitis, farriery, biomechanics, strain gauge, hoof cast
Summary
Background: Steward clogs and hoof casts are therapeutic interventions for equine laminitis, yet quantitative biomechanical evidence remains limited.
Objectives: To develop strain gauge methodology and quantify changes in principal strain and breakover time following Steward clog and hoof cast application in a horse with type I founder.
Study design: Prospective pilot study with repeated measures.
Methods: Five rosette strain gauges were positioned circumferentially on the hoof wall. Strain data were recorded at walk under three conditions: barefoot, with clog, and clog with cast. ANOVA with Tukey’s post-hoc (p<0.05) analysed data.
Results: Breakover time differed significantly between barefoot (43.2±2.1%) and clog (51.8±3.4%; p<0.001), and between clog and cast (44.1±2.8%; p<0.001). No difference existed between barefoot and cast. Total principal strain showed no significant differences (p>0.05), though regional variations occurred.
Limitations: Single subject, limited stance phases, potential gauge orientation inconsistencies.
Conclusions: Strain gauge technology successfully quantified hoof deformation. The clog prolonged breakover; the cast returned it to baseline. Overall strain reduction was not supported. Larger studies are warranted.
Introduction
Equine laminitis represents a significant clinical challenge causing pain and lameness (Pollitt, 1999). Pathophysiology involves basement membrane disruption between epidermal and dermal laminae, leading to potential distal phalanx (PIII) displacement (Pollitt and French, 2004; Dyson, 2011). Therapeutic farriery interventions include reverse shoeing (Moyer, 1980), heart bar shoes (Eustace and Caldwell, 1989), and equine digital support systems (Taylor et al., 2002). However, protocols rely primarily on anecdotal evidence rather than quantitative data (McGuigan et al., 2005).
Steward clogs combined with polymeric casts have gained clinical acceptance. Proponents suggest they reduce laminar stress through central support and modified leverage (Steward, 2003; O’Grady and Steward, 2009). Hansen et al. (2005) demonstrated casts reduce dorsal wall biaxial compression by 59%. Despite favourable case series outcomes (Steward, 2003; O’Grady et al., 2007), controlled quantitative studies remain scarce.
Strain gauge technology provides objective hoof wall deformation quantification. Thomason et al. (1992, 1998) established that hoof walls experience predominantly compressive forces during stance, with regional variations. The hoof wall functions as an anisotropic, non-homogeneous composite (Bertram and Gosline, 1987). This pilot study aimed to develop repeatable strain gauge methodology and quantify strain changes following Steward clog and hoof cast application. We hypothesized these interventions would alter strain magnitude and orientation.
Materials and Methods
Subject and Ethics
A 13-year-old mixed-breed pony (147 cm) with radiographically confirmed type I founder (Eustace, 1992) was studied with institutional approval and owner consent. Type I founder permits ethical locomotion data collection while exhibiting pathological changes. The study was non-invasive with approximately four-hour duration.
Hoof Preparation
The left forelimb was trimmed seven days prior using standardized foot mapping (Caldwell et al., 2010): heels extended palmarly to widest frog point, dorsopalmar alignment by wall dressing to phalangeal axis, mediolateral balance to live sole depth, minimal bar/frog trimming. Reference points identifying distal interphalangeal joint rotation center and biaxial toe pillars (Duckett, 1990) were marked and extended vertically for gauge placement.
Strain Gauge Application
Five encapsulated rosette gauges (1-RV91-3-120; HBM, UK) were positioned: lateral quarter (LQG), lateral toe (LTG), dorsal toe (DTG), medial toe (MTG), medial quarter (MQG). Height was standardized at 50% vertical wall height (40 mm) following Thomason et al. (1998). Each gauge had three grids (A,B,C) in stacked configuration, grid B oriented 45° to ground. Gauges were affixed with cyanoacrylate adhesive while non-weight-bearing, with 2-3 minute curing under thumb pressure. AK22 putty protected connectors. Wires (2.5 m) were secured and vet-wrapped to metacarpus/radius with joint slack.
Data Acquisition
Gauges connected to 16-channel amplifier (Quantum MX1615; HBM) powered by 7 Ah battery, linked to laptop running Catman™ software. Channels recorded principal strain 1 (E1, tensile), principal strain 2 (E2, compressive), and angles (30 channels total) at 600 Hz. All channels zeroed pre-test with limb unloaded.
Treatments
Treatment 1 (Barefoot): Post-trim baseline control.
Treatment 2 (Clog): Steward clogs (EDSS, USA) positioned using foot mapping, aligned with frog apex. Four 25 mm screws through 2 mm pilot holes. No impression material.
Treatment 3 (Cast): 75 mm Equicast™ (Equicast Inc., USA) applied over clog. Mark placed 20 mm distal to coronary band. Tape submerged 10 seconds, applied spirally, cling film wrapped during curing.
Locomotion and Analysis
Data collected at walk on tarmac: two passes per treatment (20 m straight, 10 m semicircle, 20 m straight). Five stance phases per direction randomly selected (45 total). Stance subdivided: impact, midstance, heel-off, breakover. Breakover time calculated as percentage of stance. Minitab 16® performed Anderson-Darling normality tests, Levene’s variance tests, ANOVA with Tukey’s post-hoc (p<0.05).
Results
Table 1 presents descriptive statistics. Mean stance times: 0.61-0.69 seconds. Mean E2 at breakover: -682 to -759 με.
Table 1. Principal strain 2 (E2) and temporal parameters by treatment
| Parameter | Treatment 1 (Barefoot) | Treatment 2 (Clog) | Treatment 3 (Cast) |
| Stance time (s) | |||
| Mean ± SD | 0.642 ± 0.087 | 0.687 ± 0.104 | 0.614 ± 0.093 |
| Range | 0.523-0.789 | 0.556-0.834 | 0.498-0.756 |
| Impact E2 (με) | |||
| Mean ± SD | -234 ± 87 | -198 ± 76 | -256 ± 94 |
| Midstance E2 (με) | |||
| Mean ± SD | -512 ± 134 | -487 ± 128 | -534 ± 147 |
| Breakover E2 (με) | |||
| Mean ± SD | -721 ± 156 | -682 ± 143 | -759 ± 168 |
Breakover time (% stance) | |||
| Mean ± SD | 43.2 ± 2.1 | 51.8 ± 3.4a | 44.1 ± 2.8 |
Data: mean ± SD of five gauges across three directions (n=15/treatment). E2 negative indicates compression. ap<0.001 vs Treatment 1. bp<0.001 vs Treatment 2.
Breakover time (Figure 1): barefoot 43.2±2.1%, clog 51.8±3.4% (p<0.001 vs barefoot), cast 44.1±2.8% (p<0.001 vs clog, p>0.05 vs barefoot). Principal strain at breakover (Figure 2) showed no significant differences: barefoot -721±156 με, clog -682±143 με, cast -759±168 με (p>0.05). Regional variations (Figures 3-6) revealed heterogeneous distributions varying by treatment and direction. Straight-line breakover: Treatment 1 relatively uniform (-612 to -823 με), Treatment 2 increased medial quarter strain (-891±187 με), Treatment 3 peak lateral toe (-968±201 με). Semicircle: medial quarter (inside) consistently higher (-856 to -1,024 με) versus lateral quarter (-534 to -687 με). Midstance: lower magnitude (-423 to -612 με), less heterogeneity.
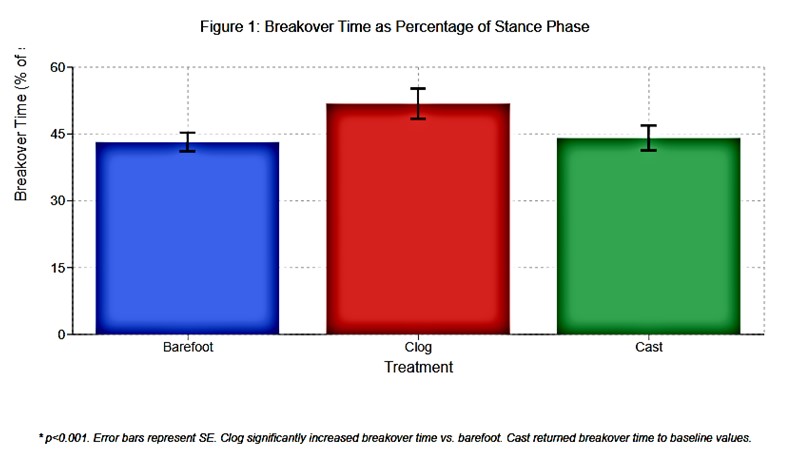
Figure 1 Histogram: Breakover time % of stance ± SE Barefoot: 43.2%, Clog: 51.8%*, Cast: 44.1%* *p<0.001
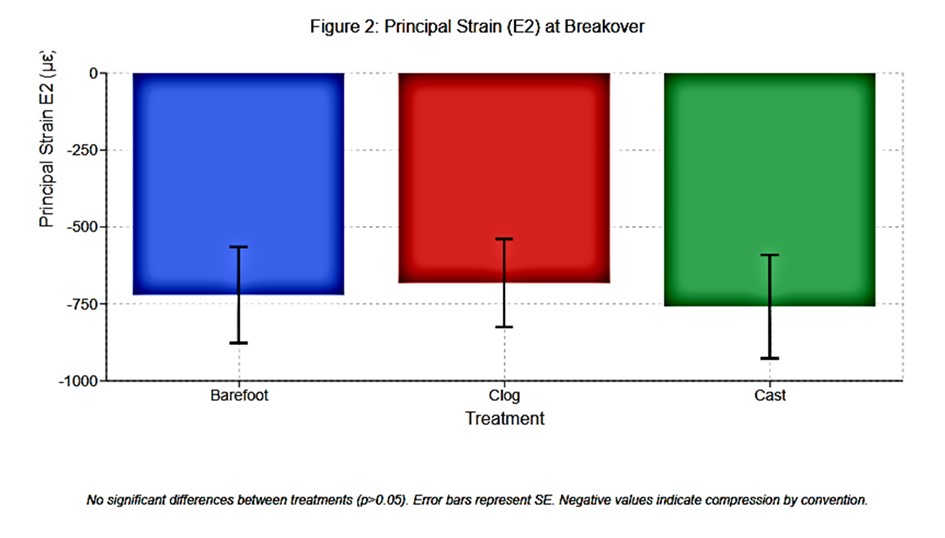
Figure 2 Histogram: Principal strain E2 at breakover ± SE Barefoot: -721 με Clog: -682 με Cast: -759 με. No significant differences.
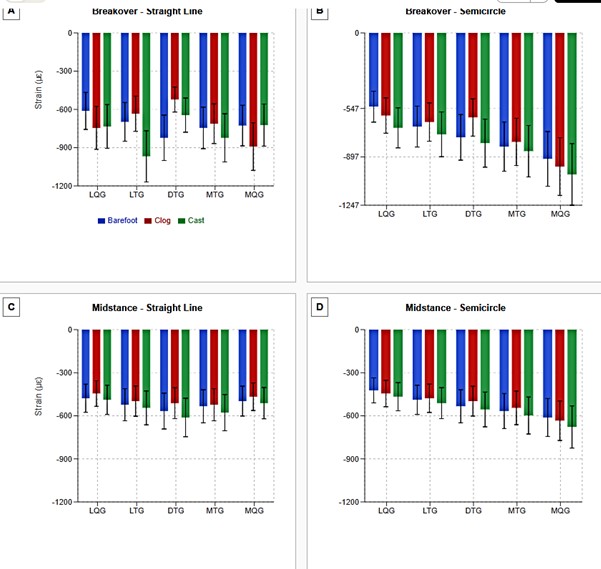
Figure 3. Regional distribution of principal strain E2 (compressive) across five-gauge locations during different locomotion conditions.
Panel A: Breakover phase during straight-line locomotion showing relatively uniform distribution in barefoot condition with regional variations in clog and cast treatments. Panel B: Breakover phase during semicircle locomotion demonstrating pronounced mediolateral asymmetry with consistently higher medial quarter (MQG) strain across all treatments, reflecting increased loading on the inside of the turn. Panel C: Midstance phase during straight-line locomotion showing lower magnitude and less heterogeneity compared to breakover. Panel D: Midstance phase during semicircle locomotion with gradual increase in strain from lateral to medial quarter.
Data presented as mean ± SD. LQG = Lateral Quarter Gauge, LTG = Lateral Toe Gauge, DTG = Dorsal Toe Gauge, MTG = Medial Toe Gauge, MQG = Medial Quarter Gauge. Blue bars = Barefoot (Treatment 1), Red bars = Clog (Treatment 2), Green bars = Cast (Treatment 3). Negative values indicate compression by convention. n = 5 stance phases per direction per treatment.
Discussion
This pilot successfully demonstrated strain gauge feasibility for quantifying hoof deformation under therapeutic interventions. The hypothesis that interventions would reduce overall strain was not supported.
Breakover Time: The striking finding was clog-induced breakover prolongation (51.8% vs 43.2% barefoot), returning to baseline with cast (44.1%). Prolonged breakover increases tensile forces on dorsal laminae during heel elevation (Kainer, 1989; Hobbs et al., 2004). The 20% increase could theoretically increase cumulative loading on damaged laminae, potentially explaining clinical discomfort during initial clog application. Cast restoration to baseline suggests it modifies mechanics beyond reinforcement. Hansen et al. (2005) showed casts reduce dorsal compression by 59%, increasing elastic modulus, potentially facilitating efficient terminal stance force transmission.
Principal Strain: Contrary to hypothesis, total strain showed no differences. This challenges clinical beliefs about dramatic strain reduction. The inherent 2° heel elevation may redistribute rather than reduce strain (Eliashar et al., 2002). Regional data support this, showing substantial location variations. Clinical improvement may result from altered distribution rather than reduction—redistributing concentrated pathological forces to healthier tissue could benefit without changing global magnitude. Measurement at 50% wall height may not capture changes at other positions. Thomason et al. (1992) demonstrated vertical gradients with maximum compression more distally.
Regional Variations: Heterogeneous distributions provide valuable insights. Pronounced mediolateral asymmetry during turns, with higher medial quarter strain, reflects expected loading (Thomason et al., 1998). Persistence across treatments suggests neither clogs nor casts alter curved locomotion demands fundamentally. Unexpected patterns deviating from normal mechanics (Thomason et al., 1992) may reflect interactions between compromised laminae, altered capsule geometry, and interventions. Peak quarter strain rather than dorsal concentration warrants investigation into whether chronic laminitis fundamentally alters load distribution pathways.
Clinical Implications: Prolonged clog-alone breakover suggests cast benefits optimize mechanics, not just retention. Clinicians should be aware breakover duration increases substantially. Regional variations highlight individualized treatment importance. Pre-treatment strain mapping might guide customization.
Limitations: Single-subject design precludes generalization. Type I founder represents mild chronic laminitis (Eustace, 1992); findings may not apply to severe cases. Limited sample size (five stances/direction/treatment) restricted statistical power. Quarter gauge placement exceeded 50% wall height due to horizontal alignment, potentially introducing variability. Poisson’s ratio suggests optimal measurement at midpoint; future studies should standardize height percentages per location or align parallel to coronary band. Encapsulated gauge stacked grids necessitated different medial/lateral orientations; non-encapsulated planar gauges (K-R81-120) would eliminate complexity. Standardized trimming may not reflect clinical variability. Radiographic guidance (O’Grady et al., 2007) and impression material (O’Grady and Steward, 2009) were omitted, potentially limiting applicability. Data limited to walk on firm surface; clinical horses experience variable conditions. Four-hour duration needs streamlining for larger studies.
Future Research: (1) Larger cohorts with varying severities would enable validation and individual variation exploration. (2) Multiple vertical gauges would characterize gradients and identify maximum benefit regions. (3) Radiographic correlation would link structural pathology with strain patterns, identifying biomechanical dysfunction predictors. (4) Systematic clog design variable evaluation (heel angle, breakover position, bevelling) would inform optimization. Comparisons with heart bars or glue-on shoes would provide relative efficacy. (5) Complete stance phase temporal analysis would comprehensively characterize mechanics. (6) Clinical outcome correlation (lameness scores, radiographic improvement, return to function) would establish whether strain predicts success. (7) Impression material quantification is essential for complete therapeutic system understanding.
Conclusions: This pilot demonstrated strain gauge feasibility for quantifying hoof biomechanics during laminitis therapy. The clog prolonged breakover; the cast returned it to baseline. Overall strain magnitude differences were not detected. Regional variations suggest therapeutic effects involve redistribution rather than global reduction. These findings provide methodological foundation for larger studies and challenge assumptions about clinical benefit mechanisms. Hoof capsule mechanics complexity in diseased states demands continued rigorous investigation to optimize interventions for this significant welfare concern.
Acknowledgments
The authors thank the horse owner, Myerscough College, Dr. J. Thomason (University of Guelph), and HBM United Kingdom Ltd.
Manufacturers’ Addresses
¹Equine Digital Support System Inc., Penrose, CO, USA
²Equicast Inc., Southern Pines, NC, USA
³HBM United Kingdom Ltd, North Harrow, Middlesex, UK
⁴Microsoft Corporation, Reading, UK
⁵Minitab Ltd, Coventry, UK
References
Bertram, J.E.A. and Gosline, J.M. (1987) Functional design of horse hoof keratin: the modulation of mechanical properties through hydration effects. Journal of Experimental Biology 130, 121-136.
Caldwell, M.N., Reilly, J.D. and Savoldi, M. (2010) Quantitative horse hoof trimming protocol for research purposes. Forge May, 4-9.
Duckett, D. (1990) The assessment of hoof symmetry and applied practical shoeing by use of an external reference point. Proceedings of the International Farriery and Lameness Seminar, Newmarket, England. Supplement 2, pp 1-11.
Dyson, S.J. (2011) Lameness and poor performance in the sport horse. In: Ross, M.W. and Dyson, S.J. (Eds.) Diagnosis and Management of Lameness in the Horse, 2nd edn., Elsevier Saunders, St Louis. pp 1108-1119.
Eliashar, E., McGuigan, M.P., Rogers, K.A. and Wilson, A.M. (2002) A comparison of three horseshoeing styles on the kinetics of breakover in sound horses. Equine Veterinary Journal 34, 184-190.
Equine Digital Support System (2006) Steward Clog Instruction Guide. Available at: http://www.hopeforsoundness.com/cms/steward-clog-instruction-guide.html [Accessed September 2013].
Eustace, R.A. (1992) Explaining Laminitis and its Prevention. Arrowsmith, Bristol.
Eustace, R.A. and Caldwell, M.N. (1989) Treatment of solar prolapse using the heart bar shoe and dorsal hoof wall resection technique. Equine Veterinary Journal 21, 370-372.
Hansen, N., Buchner, H.H.F., Haller, J. and Windischbauer, G. (2005) Evaluation using hoof wall strain gauges of a therapeutic shoe and a hoof cast with a heel wedge as potential supportive therapy for horses with laminitis. Veterinary Surgery 34, 630-636.
Hobbs, S.J., Mather, J., Rolph, C., Bower, J. and Matuszewski, B. (2004) In vitro measurement of internal hoof strain. Equine Veterinary Journal 36, 683-688.
Kainer, R.A. (1989) Clinical anatomy of the equine foot. Veterinary Clinics of North America: Equine Practice 5, 1-27.
McGuigan, M.P., Walsh, T.C., Pardoe, C.H., Day, P.S. and Wilson, A.M. (2005) Deep digital flexor tendon force and digital mechanics in normal ponies and ponies with rotation of the distal phalanx as a sequel to laminitis. Equine Veterinary Journal 37, 161-165.
Moyer, W. (1980) Corrective shoeing. Veterinary Clinics of North America: Equine Practice 2, 3-24.
O’Grady, S.E. and Steward, M.L. (2007) How to construct and apply the wooden shoe for treating three manifestations of chronic laminitis. Proceedings of the 53rd Annual Convention of the American Association of Equine Practitioners, Orlando, Florida. pp 423-429.
O’Grady, S.E. and Steward, M.L. (2009) The wooden shoe as an option for treating chronic laminitis. Equine Veterinary Education 21, 107-112.
Pollitt, C.C. (1999) Equine laminitis: a revised pathophysiology. Proceedings of the 45th Annual Convention of the American Association of Equine Practitioners, Albuquerque, New Mexico. pp 188-192.
Pollitt, C.C. and French, K.R. (2004) Equine laminitis: loss of hemidesmosomes in hoof secondary epidermal lamellae correlates to dose in an oligofructose induction model. Equine Veterinary Journal 36, 230-235.
Steward, M.L. (2003) How to construct and apply atraumatic therapeutic shoes to treat acute or chronic laminitis in the horse. Proceedings of the 49th Annual Convention of the American Association of Equine Practitioners, New Orleans, Louisiana. pp 337-346.
Taylor, D., Hood, D.M. and Wagner, I.P. (2002) Short-term effect of therapeutic shoeing on severity of lameness in horses with chronic laminitis. American Journal of Veterinary Research 63, 1629-1633.
Thomason, J.J., Biewener, A.A. and Bertram, J.E.A. (1992) Surface strain on the equine hoof wall in vivo: implications for the material design and functional morphology of the wall. Journal of Experimental Biology 166, 145-168.
Thomason, J.J. (1998) Variation in surface strain on the equine hoof wall at the midstep with shoeing, gait, substrate, direction of travel, and hoof shape. Equine Veterinary Journal Suppl. 26, 86-95.
